Abstract
Knee osteoarthritis is a degenerative joint disease, with no fundamental cure beyond pain relief and anti-inflammation. This phase I, multicenter, open-label, dose-escalation trial evaluated the safety, tolerability, and preliminary efficacy of OSCA, an intra-articular injection co-formulation of osiramestrocel (allogeneic mesenchymal stromal cells) and cartilage acellular matrix, in patients with Kellgren–Lawrence (K&L) grade 2 or 3 knee osteoarthritis. Twelve patients received a single intra-articular injection of OSCA at low (2.5 × 107 cells, n = 3), mid (5.0 × 107 cells, n = 3), or high (1.0 × 108 cells, n = 6) doses; cell concentration was 3.3 × 107 cells/ml for all cohorts, and cartilage acellular matrix was 60 mg (40 mg/ml). The primary end point was safety, assessed by dose-limiting toxicities and TEAEs. Secondary end points included Visual Analog Scale (VAS), International Knee Documentation Committee (IKDC), Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART), Whole-Organ Magnetic Resonance Imaging Score (WORMS), and K&L grading over 24 weeks. OSCA was well tolerated, with no dose-limiting toxicities and mild-to-moderate TEAEs in 25.0% (3/12) of patients. At 24 weeks, mid-dose and high-dose groups showed numerically greater improvement in pain and function than the low-dose group (P < 0.05), with up to 91.3% reduction in VAS (P = 0.002) and 102.7% IKDC (P = 0.005). MRI outcomes showed efficacy signals: MOCART total scores improved in 70% of patients, WORMS cartilage integrity improved in 50%, and K&L grades remained stable in 80%. Overall, these findings support the feasibility and tolerability of a single OSCA injection, with exploratory efficacy in both clinical and MRI outcomes.
Similar content being viewed by others
Introduction
Osteoarthritis is a degenerative joint disease characterized by progressive cartilage loss, subchondral bone remodeling, synovial inflammation, and joint pain, ultimately leading to functional impairment and disability1,2. Osteoarthritis, a leading cause of chronic pain and disability worldwide, sustantially impairs the quality of life of patients and imposes a major socioeconomic burden3,4. Despite its prevalence, osteoarthritis still lacks treatments that modify disease progression or restore joint function. Current therapies — analgesics, non-steroidal anti-inflammatory drugs, and intra-articular (IA) injections — mainly target symptomatic relief, as no disease-modifying osteoarthritis drug (DMOAD) has been approved5,6,7.
Recently, mesenchymal stem cell (MSC)-based therapies have demonstrated potential for cartilage protection and regeneration in osteoarthritis8,9,10,11,12. However, structural recovery in the knee joint, such as cartilage repair and subchondral bone regions, has not been consistently demonstrated in clinical studies. Among them, studies using MSCs have shown either no meaningful changes in bone marrow lesions or cartilage morphology, or only limited evidence of cartilage regeneration in short-term follow-up13,14,15. Among various MSC sources, human umbilical cord blood-derived MSCs stand out as a promising regenerative therapy for osteoarthritis owing to their high proliferative capacity, low immunogenicity, and secretion of bioactive factors that facilitate cartilage repair and modulate the joint microenvironment11,12,16. Therefore, to date, no approved DMOAD effectively stops disease progression, preserves joint structure, and improves physical function beyond pain relief.
OSCA is a novel therapy, a co-formulation of OSiramestrocel (human umbilical cord blood-derived allogeneic mesenchymal stromal cells) and Cartilage Acellular matrix (CAM) that enhances cartilage regeneration through their synergistic interaction. As small animal models such as rabbits and rats have excellent self-healing ability, it is difficult to expect equivalent efficacy in actual clinical practice17,18. Therefore, for the clinical application, it is required to evaluate the therapeutic efficacy, stability, and biological safety in large animal models that have less self-healing ability and better mimic the anatomical features of human articular joints19,20,21. Our previous study demonstrated that IA OSCA injections sustantially promoted cartilage repair, modulated the joint microenvironment, reduced inflammation, and supported subchondral bone remodeling, which highlighted their potential as a DMOAD22,23. Therefore, this first-in-human phase I study aims to evaluate the safety and tolerability of OSCA and its potential disease-modifying effects on joint structure in patients with knee osteoarthritis.
Methods
Study design and patient eligibility
This study is a first-in-human, multicenter, open-label clinical trial designed to evaluate the safety, tolerability, and clinical efficacy of OSCA. Patient enrollment began on July 11, 2023, and the last patient visit was conducted on August 6, 2024. The clinical trial adhered to the guidelines of the Declaration of Helsinki and was approved by the Institutional Review Board (IRB) of Kyung Hee University Hospital and Kyung Hee University Hospital at Gangdong (IRB no. 2023-01-073-004 and 2023-01-008) and registered at ClinicalTrials.gov (NCT05944627). Written informed consent was obtained from all patients before enrollment.
Eligible patients were adults (\(\ge\)19 years old) diagnosed with knee osteoarthritis based on standard weight-bearing radiographs showing Kellgren–Lawrence (K&L) grade 2–3 and confirmed by the International Cartilage Repair Society (ICRS) grade 3 or 4 rating system through MRI examination at the time of screening. Additionally, participants were required to have a Visual Analog Scale (VAS) pain score of \(\ge\)50 mm on a 100-mm scale at screening and persistent symptoms (pain, and so on) that do not improve after at least 12 weeks of conservative treatment (drug therapy, physical therapy, and so on). Detailed inclusion and exclusion criteria are provided (Supplementary Table 1).
Enrollment and study protocol
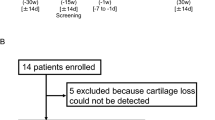
Following eligibility assessment, patients were sequentially enrolled and administered a single IA injection of OSCA at one of three escalating doses. A single IA injection containing 2.5 \(\times\) 107 cells (cohort 1, low-dose), 5.0 \(\times\) 107 cells (cohort 2, mid-dose), or 1.0 \(\times\) 108 cells (cohort 3, high-dose), with a cell concentration of 3.3 \(\times\) 107 cells/ml for all cohorts, combined with CAM 60 mg at a concentration of 40 mg/ml, was administered. Dose escalation followed a standard 3 \(+\) 3 design based on dose-limiting toxicities (DLTs). Patients were followed up on day 1 and weeks 1, 4, 8, 12, 16, 20, and 24 post-injection to assess safety and exploratory efficacy using pain and functional scores. Additionally, radiological end points were evaluated at baseline and 24 weeks (Fig. 1). During the clinical trial period, Celecoxib (Celebrex® 100 mg capsules) was permitted as rescue medication for pain management at a maximum daily dose of 200 mg, administered either as a single 200 mg dose or 100 mg twice daily. However, to ensure accurate efficacy assessments, the administration of rescue medications was discontinued at least 5 days before the scheduled evaluation visit. In addition, the use of non-steroidal anti-inflammatory drugs outside the permitted concomitant drugs, including analgesics, systemic corticosteroids, immunosuppressants, muscle relaxants, and IA therapies, was prohibited and could lead to participant discontinuation.
This figure illustrates the study design, including participant allocation, follow-up schedule, and assessment timeline. Fourteen patients with knee osteoarthritis were screened and allocated to three cohorts: low-dose (n = 3), middle-dose (n = 3), and high-dose (n = 6). Two patients from the high-dose cohort discontinued participation owing to contraindicated medication use (n = 1) and adverse events (n = 1). All remaining participants completed follow-up assessments over 24 weeks. Assessments were conducted on day 1 and at weeks 1, 4, 8, 12, 16, 20, and 24 post-intra-articular OSCA administration to assess safety and exploratory efficacy via pain and function scores. Radiological exploratory end points were evaluated at baseline and at 24 weeks, whereas biomarker analyses were conducted at baseline and weeks 1, 4, 12, and 24. DLT, dose-limiting toxicity.
OSCA mechanism of action and intervention
The therapeutic efficacy of the OSCA intervention is mediated through modulation of the joint microenvironment. By restoring the balance between catabolic and anabolic signaling pathways, OSCA promotes tissue homeostasis within the osteoarthritic joint. This biological effect is driven by three synergistic mechanisms: stimulation of chondrogenic differentiation, which facilitates endogenous cartilage repair, immunomodulation, resulting in the attenuation of inflammatory responses, and protective effect through the inhibition of apoptosis in joint-resident cell. Collectively, these processes contribute to structural preservation of articular cartilage and improvement in overall joint function (Supplementary Fig. 1).
The frozen osiramestrocel and CAM components were thawed immediately before administration and aseptically combined into a single syringe. The injection site was aseptically prepared, and the solution was administered into the knee joint via the superolateral aspect by an orthopedic specialist uninvolved in patient assessment. Following administration, patients were closely monitored for 30 min for any adverse events, including injection site reactions.
Biomarker measurement and sample collection
Urinary CTX-I and CTX-II were measured using ELISA kits (IDS, AC-03F1R and AC-10F1R; Boldon, UK) and normalized to urinary creatinine. Serum levels of CTX-I (IDS, AC-02F1R; Boldon), CTX-II (USCNK, CEA686Hu; Wuhan, China), MMP-3 (R&D Systems, DMP300; Minneapolis, MN, USA), PIIANP (MyBioSource, MBS109368; San Diego, CA, USA), and COMP (IDS, AC-23F1R; Boldon) were quantified via ELISA. Serum IL-1β and tumor necrosis factor-alpha (TNF-α) were assessed using a MILLIPLEX MAP Human High Sensitivity T Cell Panel-Immunology Multiplex Assay (Merck, HSTCMAG-28SK; Darmstadt, Germany). Urine and blood samples were collected at baseline and weeks 1, 4, 12, and 24. Midstream first-morning urine was stored at \(\le\)−70 °C. Blood samples were left at room temperature for 30 min, then centrifuged at 3,000 rpm for 10 min, and the serum aliquots were stored at \(\le\)−70 °C until analysis.
Accuracy assessment
In each analytical run, calibration samples were analyzed in duplicate, and accuracy (% recovery) was evaluated using the mean value. If accuracy exceeded the acceptance criteria, the individual value was used instead.
Outcome measurement
The primary end point was to assess DLT and to determine maximum tolerated dose after a single IA OSCA injection in patients with knee osteoarthritis. Safety and tolerability were evaluated based on the incidence of DLT, treatment-emergent adverse events (TEAEs), adverse drug reactions, and serious adverse events across all cohorts. The maximum tolerated dose was determined by monitoring DLT frequency and severity within a dose-escalation design, with adverse events classified and graded as per the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE ver.5.0)24.
Secondary end points assessed exploratory efficacy after administration of OSCA over 24 weeks based on changes in pain and function scores using the International Knee Documentation Committee (IKDC)25, Knee Injury and Osteoarthritis Outcome Score (KOOS)26, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC)27, and VAS28 indices. Higher IKDC and KOOS scores indicate better clinical outcomes, whereas lower WOMAC and VAS scores denote symptom relief. Additional analysis of clinical efficacy was conducted to identify responders and non-responders using prespecified minimal clinically important difference-based criteria; responders were defined as those with a high improvement in pain or function (\(\ge\)50% relative improvement and \(\ge\)20-point absolute change from baseline)29. In addition, joint structural changes were evaluated using MRI and radiography to investigate the potential disease-modifying effects of OSCA. The Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) 2.0 score30 and Whole-Organ Magnetic Resonance Imaging Score (WORMS)31 were used for MRI assessment, whereas the K&L grading was used to evaluate radiographs. MOCART and WORMS assessments were performed on 3T MR systems (Achieva and Ingenia, Philips Medical Systems, Eindhoven, The Netherlands) at the baseline and 24 weeks after a single IA OSCA injection. Supplementary Table 2 shows the detailed MRI sequence parameters. An increased MOCART score indicated cartilage repair, whereas a decreased WORMS score and K&L grade indicated improved joint structure. All MRI and X-ray images were uploaded to a centralized clinical trial imaging management system (Trial Informatics, Seoul, Korea) and blindly evaluated by two independent radiologists. If the two primary readers disagreed, an adjudicating radiologist (third independent reader) reviewed their assessment scores and selected the most accurate result without conducting a separate evaluation. Additionally, exploratory biomarker analyses were conducted over 24 weeks to investigate the biological effects of OSCA on cartilage degradation and inflammation.
Statistical analysis
Continuous variables are reported as mean and standard deviation, whereas categorical variables are reported as frequency and percentage. All end point analyses were conducted based solely on observed data, with no imputation performed for missing values. After normality assessment, either the Kruskal–Wallis test or one-way analysis of variance was used for all cohort comparisons of continuous variables, whereas the Mann–Whitney U test or Tukey’s honestly significant difference was used for post hoc pairwise comparisons as appropriate. Categorical variables were analyzed using Pearson’s χ or Fisher’s exact test, and within-cohort changes from baseline to 24 weeks were analyzed using the Wilcoxon signed-rank test. To account for within-subject correlation owing to repeated measurements and to evaluate longitudinal changes over time, a linear mixed-effect model was utilized to assess cohort differences over the 24-week follow-up period. The model included patients as random effects and cohorts, visit time, and their interactions as fixed effects. Post hoc pairwise comparisons were conducted to identify significant differences between cohorts. For clinical interpretability, responder proportions were compared across cohorts using Fisher’s exact test, based on predefined clinically meaningful thresholds corresponding to the minimal clinically important difference for pain and function. Data were analyzed with SAS Version 9.4 (SAS Institute, Cary, NC, USA). A two-sided P-value <0.05 indicated statistical significance.
Results
Baseline characteristics
Twelve patients were sequentially assigned to one of three dose cohorts: low-dose (n = 3), mid-dose (n = 3), and high-dose (n = 6). During the 24-week follow-up period, two patients in the high-dose group discontinued participation — one due to prohibited concomitant medication use and one due to adverse events. Ultimately, 10 patients completed the study. Baseline demographic and clinical measures (age, sex, body mass index, pain, and function) were similar across all three groups without statistically significant differences. Table 1 presents the detailed baseline characteristics.
Safety and tolerability
IA administration of OSCA was well tolerated across all cohorts, showing a favorable safety profile (Table 2). No DLTs or TEAEs causing death were reported throughout the study. TEAEs occurred in 25.0% (3/12 patients), with a total of four events, mostly mild-to-moderate (grades 2–3). The reported TEAEs were nasopharyngitis (cohort 2; n = 1), dyspepsia (cohort 3; n = 1), knee swelling (cohort 3; n = 1), and transverse myelitis (cohort 3, n = 1). An adverse drug reaction (knee swelling) occurred in cohort 3 (8.3%) and resolved within 6 weeks. In addition, one SAE (acute transverse myelitis) occurred in cohort 3 (8.3%) but was unrelated to the investigational product. Two patients in three cohorts discontinued owing to protocol deviations, including prohibited medication use and adverse events that prevented continued participation.
Clinical outcomes for pain and function
IKDC scores increased from baseline (cohort 1: 37.55 \(\pm\) 14.04; cohort 2: 41.38 \(\pm\) 11.32; cohort 3: 40.52 \(\pm\) 8.63) to 24 weeks (cohort 1: 42.91 \(\pm\) 14.55; cohort 2: 70.88 \(\pm\) 17.45; cohort 3: 79.02 \(\pm\) 13.61), with mean changes of \(+\)5.36, \(+\)29.50, and \(+\)38.50 scores, corresponding to mean change rates of \(+\)15.3%, \(+\)89.7%, and \(+\)102.7%, respectively. WOMAC scores decreased from baseline (cohort 1: 44.33 \(\pm\) 10.02; cohort 2: 50.00 \(\pm\) 17.35; cohort 3: 31.00 \(\pm\) 13.37) to 24 weeks (cohort 1: 42.00 \(\pm\) 8.19; cohort 2: 13.67 \(\pm\) 15.82; cohort 3: 5.00 \(\pm\) 4.97), with mean changes of \(-\)2.33, \(-\)36.33, and \(-\)26.00 scores (mean change rates of \(-\)4.7%, \(-\)60.5%, and \(-\)76.2%, respectively). Similarly, VAS scores also decreased from baseline (cohort 1: 70.00 \(\pm\) 7.00; cohort 2: 77.00 \(\pm\) 13.23; cohort 3: 69.25 \(\pm\) 13.05) to 24 weeks (cohort 1: 59.67 \(\pm\) 8.14; cohort 2: 14.00 \(\pm\) 17.06; cohort 3: 5.25 \(\pm\) 5.50), with mean changes of \(-\)10.33, \(-\)63.00, and \(-\)64.00 scores (mean change rates of \(-\)14.6%, \(-\)78.9%, and \(-\)91.3%, respectively). All KOOS subscales, including pain, symptoms, stiffness, activities of daily living, sport and recreation function, and quality of life, improved across cohorts. Detailed numerical results for secondary end points are provided in Table 3 and Fig. 2.
Mean changes over 24 weeks in Visual Analog Scale, total Western Ontario and McMaster Universities Osteoarthritis Index score, International Knee Documentation Committee, and Knee Injury and Osteoarthritis Outcome Score subscales (pain, symptoms, activities of daily living, sports and recreation function, and quality of life) for patients receiving low-dose (blue), mid-dose (green), or high-dose (orange) OSCA.
Following IA administration of OSCA, exploratory linear mixed-effect model analysis based on absolute change scores indicated that cohorts 2 and 3 showed numerically greater improvement in pain and functional outcomes over 24 weeks compared with cohort 1. Specifically, greater numerical improvements were observed in VAS (P = 0.002) and IKDC (P = 0.005) scores for cohort 3 and in WOMAC scores (P = 0.028) for cohort 2, compared with cohort 1. For the KOOS subscales, greater numerical improvements were noted in pain (P = 0.011), symptoms (P = 0.007), activities of daily living (P = 0.041), and quality of life (P = 0.004) for cohort 2 and in sports and recreation function (P = 0.021) for cohort 3, compared with cohort 1 (Table 3). The responder proportions, defined a priori using a clinically meaningful threshold, are summarized across VAS, WOMAC, IKDC, and KOOS subscales in Table 4. No responders were observed in the low-dose cohort across measures. By contrast, the high-dose cohort showed a 100% responder rate (4/4 patients) for the VAS, and a 75% rate (3/4 patients) was observed for the WOMAC, IKDC, and KOOS subscale (quality of life) (Table 4).
Radiological outcomes for joint structural changes
Across OSCA-treated cohorts, the mean MOCART score increased over 24 weeks, with seven patients (70%) showing improvement; however, this increase was not statistically significant (P > 0.05) (Supplementary Table 3).
Nine out of 10 (90%) patients maintained structural stability in subchondral bone; 8 (80%) showed no structural progression, and 1 (10%) improved (Fig. 3). Subchondral bone remodeling was observed exclusively in cohort 3, affecting one of the four patients (25%), with evidence of improved bone marrow and structural modification (Fig. 4). Additionally, cartilage defect volume fill improved in six patients (60%) across all cohorts.
a Percentage distribution of MOCART total scores and subscore changes at 24 weeks following intra-articular OSCA injection. Improvements (green), stability (light green), and worsening (grey). Across cohorts, 70% of patients showed improvement in MOCART scores, indicating potential cartilage repair effects. b WORMS total scores and subcomponent changes at 24 weeks across cohorts, with improvements (green), stability (light green), and worsening (grey). WORMS total score improved in 40% of patients, and cartilage integrity improved in 50% based on cartilage assessment subscores. Bone marrow abnormality scores were stable or improved in most participants, suggesting potential joint preservation effects of OSCA. MOCART, Magnetic Resonance Observation of Cartilage Repair Tissue; WORMS, Whole-Organ Magnetic Resonance Imaging Score.
a Sagittal MRI at baseline shows cartilage defects in the lateral femoral condyle (yellow arrow) and lateral tibial plateau (red arrow). b At 24 weeks, MRI revealed increased cartilage coverage in the lateral femoral condyle (yellow arrow) and lateral tibial plateau (red arrow) with surface smoothening. c Baseline sagittal MRI images show subchondral bone marrow edema (red arrows) in the medial femoral condyle. d At 24 weeks, subchondral bone marrow integrity improved (red arrow).
Although the mean WORMS score showed no statistically significant changes from baseline to 24 weeks (Supplementary Table 3), four patients (40%) showed a reduction in WORMS total score. Five patients (50%) showed a reduction in the cartilage assessment component of WORMS (Figs. 3 and 4). However, osteophyte score worsened in seven patients (70%). Among the 10 participants, 2 (20%) exhibited a progression in K&L grade (one from grade 2 to grade 3 and one from grade 3 to grade 4), whereas 8 (80%) remained unchanged.
Biomarker outcomes
Following OSCA administration, inflammation-related biomarkers tended to decrease over 24 weeks; however, none of the within-groups change from baseline to 24 weeks reached statistical significance. Serum IL-1β and TNF-α levels showed a downward trend over 24 weeks in the low-dose and medium-dose groups. Serum PIIANP tended to increase in the medium-dose and high-dose groups but decrease in the low-dose group, with no significant changes. Urinary CTX-II remained stable, whereas serum CTX-II showed a gradual, non-significant increase. Serum COMP and MMP-3 remained stable across all groups over 24 weeks (Supplementary Fig. 2).
Discussion
This first-in-human, multicenter phase I study suggested that IA administration of OSCA might be a potential and innovative treatment for knee osteoarthritis, demonstrating a favorable efficacy and well-tolerated safety profile in low-dose, mid-dose, and high-dose of OSCA. In addition to symptomatic relief, the inclusion of both patient-reported outcomes and MRI-based scoring systems, such as MOCART and WORMS, allowed for a more structured evaluation of treatment effects on joint integrity. With respect to clinical relevance, structural changes on MRI such as cartilage thickness typically require longer follow-up periods often on the order of 2 years to demonstrate robust between-group differences in osteoarthritis trials. For instance, the FORWARD randomized clinical trial of IA sprifermin, the primary structural end point (change in total femorotibial joint cartilage thickness measured by quantitative MRI) was assessed at 2 years32. Although our MRI assessment relied on semi-quantitative scoring rather than quantitative thickness, directional changes in cartilage-related components may be clinically relevant; however, this phase I study was not powered to determine imaging–symptom relationships. The observation of osteophyte progression in some participants highlights the heterogeneity of structural remodeling and the need for longer-term controlled studies to clarify clinical significance. Taken together, observed clinical improvements and imaging-based structural changes suggest that OSCA may exert effects beyond symptom control and potentially influence the underlying disease process. Longer-term follow-up is currently ongoing to further validate these findings.
Several randomized controlled trials have evaluated IA MSC injections using WOMAC and WORMS, but a meta-analysis revealed limited evidence for pain relief and functional improvement, as well as insufficient evidence for cartilage repair33. Studies report that UCB-MSC-HA implants improve VAS, WOMAC, and IKDC scores10,34, but their use remains limited to patients receiving adjunctive treatment with microfracture for ICRS grade IV cartilage defects and not in the general osteoarthritis population. To our knowledge, no therapy is currently approved as a DMOAD that regenerates cartilage and modifies joint structure, underscoring the unmet need in knee osteoarthritis treatment. Most regenerative approaches target localized defects35,36, whereas our phase I data suggest that OSCA may promote symptomatic improvement with variable structural findings on MRI, warranting confirmation in further controlled trials. A notable strength of OSCA lies in its combination therapy strategy, in which CAM serves as a supportive adjunct to novel approach designed to enhance therapeutic efficacy by augmenting the activity of MSCs and promoting their regenerative capacity22,23. The numerical improvement in the volume fill of cartilage defect subscores in MOCART following OSCA administration may be mainly attributable to the optimized microenvironment created by OSCA, which could facilitate cartilage regeneration. This component enhances the local microenvironment to promote stem cell chondrogenic differentiation and restores essential cartilage components, such as sulfated glycosaminoglycans and collagen, thereby contributing to effective tissue repair17,22. Cartilage tissue improvement after OSCA administration aligns with the findings of previous preclinical studies22,23, further supporting their translational relevance.
Serum IL-1β and TNF-α, pro-inflammatory cytokines related to joint inflammation and cartilage degradation18,19, showed a non-significant decreasing trend up to 24 weeks in the low-dose and mid-dose cohorts after OSCA administration, suggesting a possible reduction in inflammatory burden in this exploratory phase I study. This trend may be attributable to the immunomodulatory properties of MSCs and the supportive microenvironment provided by CAM, which together may modulate IA inflammation. Serum PIIANP, a marker of cartilage synthesis20, tended to increase in the mid-dose and high-dose groups; however, in the absence of statistical correlation with clinical outcomes, these findings should be interpreted cautiously and considered hypothesis-generating.
WORMS analysis revealed that some patients exhibited osteophyte formation following OSCA administration. IA injection of stem cells activates key growth factors, such as TGF-βs and bone morphometric proteins, that have pivotal roles in joint tissue regeneration21,37,38. Notably, both TGF-βs and bone morphometric proteins have also been implicated in osteophyte formation within the joint39,40,41. Therefore, the observed osteophyte changes after OSCA administration may reflect activation of joint remodeling processes rather than definitive disease progression. Given the exploratory nature of this phase I study and the limited follow-up duration, osteophyte changes should be interpreted with caution and warrant long-term monitoring to clarify their clinical significance.
This study has some limitations. First, the small sample size of only 12 participants limits statistical power, generalizability, and the interpretability of inferential statistics; therefore, efficacy signals from secondary outcomes should be interpreted cautiously as exploratory. Second, the 24-week follow-up limited the assessment of long-term efficacy and safety. To address this limitation, a 5-year long-term follow-up study (NCT06013306) is currently ongoing and is expected to provide more definitive data on the long-term efficacy and safety of OSCA. Third, the study population included only Korean patients, which may limit the generalizability of the results to other ethnic backgrounds, especially with regard to body mass index. Finally, our study was not designed as a randomized, double-blind trial, which may introduce assessment biases and limit the strength of our findings. Despite limitations, our phase I clinical trial study provides valuable preliminary evidence supporting the feasibility, safety, and potential therapeutic efficacy of IA OSCA administration for knee osteoarthritis.
Over 24 weeks, numerical changes in pain and function outcomes were observed, and responder analyses based on a clinically meaningful threshold provided clinically interpretable context, although between-cohort comparisons should be considered exploratory given the small cohort sizes. MRI-based assessments showed mixed findings; these results should be considered hypothesis-generating and require confirmation in larger randomized controlled trials. These early-phase findings warrant further evaluation of OSCA as a potential DMOAD for knee osteoarthritis. To this end, a phase IIa trial involving 108 patients has commenced to further evaluate the efficacy and safety of OSCA as part of broader, long-term, large-scale clinical investigations.
References
Barnett, R. Osteoarthritis. Lancet 391, 1985 (2018).
Li, S., Cao, P., Chen, T. & Ding, C. Latest insights in disease-modifying osteoarthritis drugs development. Ther Adv Musculoskelet Dis. 15, 1759720x231169839 (2023).
Hunter, D. J. & Bierma-Zeinstra, S. Osteoarthritis. Lancet 393, 1745–1759 (2019).
Litwic, A., Edwards, M. H., Dennison, E. M. & Cooper, C. Epidemiology and burden of osteoarthritis. Br. Med. Bull. 105, 185–199 (2013).
Sharma, L. Osteoarthritis of the knee. N. Engl. J. Med. 384, 51–59 (2021).
Singh, H. et al. Relative efficacy of intra-articular injections in the treatment of knee osteoarthritis: a systematic review and network meta-analysis. Am. J. Sports Med. 50, 3140–3148 (2022).
Bannuru, R. R. et al. OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis. Osteoarthr. Cartil. 27, 1578–1589 (2019).
Chanda, D., Kumar, S. & Ponnazhagan, S. Therapeutic potential of adult bone marrow-derived mesenchymal stem cells in diseases of the skeleton. J. Cell. Biochem. 111, 249–257 (2010).
Zhang, R., Ma, J., Han, J., Zhang, W. & Ma, J. Mesenchymal stem cell related therapies for cartilage lesions and osteoarthritis. Am. J. Transl. Res. 11, 6275–6289 (2019).
Park, Y. B., Ha, C. W., Lee, C. H., Yoon, Y. C. & Park, Y. G. Cartilage regeneration in osteoarthritic patients by a composite of allogeneic umbilical cord blood-derived mesenchymal stem cells and hyaluronate hydrogel: results from a clinical trial for safety and proof-of-concept with 7 years of extended follow-up. Stem Cells Transl. Med. 6, 613–621 (2017).
Lee, D. H. et al. Cartilage regeneration using human umbilical cord blood derived mesenchymal stem cells: asystemati c review and meta-analysis. Medicina (Kaunas) 58,1801 (2022).
Lee, N. H. et al. Allogenic human umbilical cord blood-derived mesenchymal stem cells are more effective than bone marrow aspiration concentrate for cartilage regeneration after high tibial osteotomy in medial unicompartmental osteoarthritis of knee. Arthroscopy 37, 2521–2530 (2021).
Matas, J. et al. Umbilical cord-derived mesenchymal stromal cells (MSCs) for knee osteoarthritis: repeated MSC dosing is superior to a single MSC dose and to hyaluronic acid in a controlled randomized phase I/II trial. Stem Cells Transl. Med. 8, 215–224 (2019).
Kim, K. I., Kim, M. S. & Kim, J. H. Intra-articular injection of autologous adipose-derived stem cells or stromal vascular fractions: are they effective for patients with knee osteoarthritis? A systematic review with meta-analysis of randomized controlled trials. Am. J. Sports Med. 51, 837–848 (2023).
Higuchi, J. et al. Associations of clinical outcomes and MRI findings in intra-articular administration of autologous adipose-derived stem cells for knee osteoarthritis. Regen. Ther. 14, 332–340 (2020).
Kim, J. Y., Jeon, H. B., Yang, Y. S., Oh, W. & Chang, J. W. Application of human umbilical cord blood-derived mesenchymal stem cells in disease models. World J. Stem Cells 2, 34–38 (2010).
Yang, G. et al. Transplantation of human umbilical cord blood-derived mesenchymal stem cells improves cartilage repair in a rabbit model. Biomed. Res. Int. 2021, 6380141 (2021).
Molnar, V. et al. Cytokines and chemokines involved in osteoarthritis pathogenesis. Int. J. Mol. Sci. 22, 9208 (2021).
Yang, J. et al. IL‑1β increases the expression of inflammatory factors in synovial fluid‑derived fibroblast‑like synoviocytes via activation of the NF‑κB‑mediated ERK‑STAT1 signaling pathway. Mol. Med. Rep. 20, 4993–5001 (2019).
Kraus, V. B. & Karsdal, M. A. Osteoarthritis: current molecular biomarkers and the way forward. Calcif. Tissue Int. 109, 329–338 (2021).
Deng, Z. H., Li, Y. S., Gao, X., Lei, G. H. & Huard, J. Bone morphogenetic proteins for articular cartilage regeneration. Osteoarthr. Cartil. 26, 1153–1161 (2018).
Kim, M. et al. Combined mesenchymal stem cells and cartilage acellular matrix injection therapy for osteoarthritis in goats. Tissue Eng. Regen. Med. 19, 177–187 (2022).
Jeon, H. J. et al. Therapeutic effects of human umbilical cord blood-derived mesenchymal stem cells combined with cartilage acellular matrix mediated via bone morphogenic protein 6 in a rabbit model of articular cruciate ligament transection. Stem Cell Rev Rep. 16, 596–611 (2020).
Basch, E. et al. Patient versus clinician symptom reporting using the National Cancer Institute Common Terminology Criteria for Adverse Events: results of a questionnaire-based study. Lancet Oncol 7, 903–909 (2006).
Kim, J. G. et al. Translation and validation of the Korean version of the International Knee Documentation Committee subjective knee form. Knee Surg. Relat. Res. 25, 106–111 (2013).
Roos, E. M., Roos, H. P., Lohmander, L. S., Ekdahl, C. & Beynnon, B. D. Knee Injury and Osteoarthritis Outcome Score (KOOS) — development of a self-administered outcome measure. J. Orthop. Sports Phys. Ther. 28, 88–96 (1998).
Bellamy, N., Buchanan, W. W., Goldsmith, C. H., Campbell, J. & Stitt, L. W. Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J. Rheumatol. 15, 1833–1840 (1988).
Hawker, G. A., Mian, S., Kendzerska, T. & French, M. Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF-36 BPS), and Measure of Intermittent and Constant Osteoarthritis Pain (ICOAP). Arthritis Care Res. (Hoboken) 63, S240–S252 (2011).
Pham, T. et al. OMERACT-OARSI initiative: Osteoarthritis Research Society International set of responder criteria for osteoarthritis clinical trials revisited. Osteoarthr. Cartil. 12, 389–399 (2004).
Schreiner, M. M. et al. The MOCART (Magnetic Resonance Observation of Cartilage Repair Tissue) 2.0 Knee Score and Atlas. Cartilage 13, 571s–587s (2021).
Peterfy, C. G. et al. Whole-Organ Magnetic Resonance Imaging Score (WORMS) of the knee in osteoarthritis. Osteoarthr Cartil. 12, 177–190 (2004).
Hochberg, M. C. et al. Effect of intra-articular sprifermin vs placebo on femorotibial joint cartilage thickness in patients with osteoarthritis: the FORWARD randomized clinical trial. JAMA 322, 1360–1370 (2019).
Kim, S. H. et al. Intra-articular injection of mesenchymal stem cells for clinical outcomes and cartilage repair in osteoarthritis of the knee: a meta-analysis of randomized controlled trials. Arch. Orthop. Trauma Surg. 139, 971–980 (2019).
Lim, H. C. et al. Allogeneic umbilical cord blood-derived mesenchymal stem cell implantation versus microfracture for large, full-thickness cartilage defects in older patients: a multicenter randomized clinical trial and extended 5-year clinical follow-up. Orthop. J. Sports Med. 9, 2325967120973052 (2021).
Barry, F. & Murphy, M. Mesenchymal stem cells in joint disease and repair. Nat. Rev. Rheumatol. 9, 584–594 (2013).
Wang, A. T., Feng, Y., Jia, H. H., Zhao, M. & Yu, H. Application of mesenchymal stem cell therapy for the treatment of osteoarthritis of the knee: a concise review. World J. Stem Cells 11, 222–235 (2019).
Tian, R. et al. Revolutionizing osteoarthritis treatment: how mesenchymal stem cells hold the key. Biomed. Pharmacother. 173, 116458 (2024).
Yoo, K. H. et al. Transforming growth factor‑β family and stem cell‑derived exosome therapeutic treatment inosteoart hritis (Review). Int. J. Mol. Med. 49, 62 (2022).
Blaney Davidson, E. N., van der Kraan, P. M. & van den Berg, W. B. TGF-beta and osteoarthritis. Osteoarthr. Cartil. 15, 597–604 (2007).
Blaney Davidson, E. N. et al. Resemblance of osteophytes in experimental osteoarthritis to transforming growth factor beta-induced osteophytes: limited role of bone morphogenetic protein in early osteoarthritic osteophyte formation. Arthritis Rheum 56, 4065–4073 (2007).
van Beuningen, H. M., van der Kraan, P. M., Arntz, O. J. & van den Berg, W. B. Transforming growth factor-beta 1 stimulates articular chondrocyte proteoglycan synthesis and induces osteophyte formation in the murine knee joint. Lab. Invest. 71, 279–290 (1994).
Acknowledgements
This research was supported by the Korean Fund for Regenerative Medicine grant funded by the Korea government (the Ministry of Science and ICT, the Ministry of Health and Welfare) (Grant No. RS-2025-02122977).
Funding
This research was supported by the Korean Fund for Regenerative Medicine grant funded by the Korea government (the Ministry of Science and ICT, the Ministry of Health and Welfare) (Grant No. RS-2025-02122977).
Author information
Authors and Affiliations
Contributions
H.-J.K., Y.B., J.C.N., and K.-S.K. contributed to the planning and the design of the clinical trials. S.H.L., M.K., and K.-S.K provided critical discussion. D.K.S., S.H.L., K.-S.K., and K.H.Y wrote the main manuscript. All authors contributed with study design, decision to publish, and preparation of the manuscript. All authors have reviewed and approved the final version of this manuscript.
Corresponding authors
Ethics declarations
Competing interests
H.-J.K., Y.B., S.H.L., M.K., and J.C.N. are employees of Kangstem Biotech Co., Ltd, and K.-S.K. is the Founder of Kangstem Biotech Co., Ltd. This commercial affiliation did not have any role in the data collection and analysis. All other authors declare no conflicts of interest in the authorship or publication of this contribution.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Suh, D.K., Lee, S.H., Bae, Y. et al. First-in-human and multicenter phase I study of OSCA therapy for knee osteoarthritis. Exp Mol Med (2026). https://doi.org/10.1038/s12276-026-01728-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s12276-026-01728-w